Ideal Incubation Conditions
Cells and tissues cultivated in vitro simply need the same conditions as they have in vivo. Easily said and easy to understand, it is often difficult to achieve this on a microscopic setup. With our long-term experience in microscopic cell and tissue cultivation, we have developed solutions that are both easy to use and give the cells the best possible, most homogeneous conditions in every incubation-relevant parameter.
Temperature
Temperature is the primary parameter for a successful in vitro cell cultivation. If the temperature is not like in vivo, a differing physiological behavior will occur very rapidly. Current scientific research is ongoing in this field, and a lot of temperature sensitive proteins have already been identified. Since the first observation of the expression of specific proteins was under heat stress, they are generally called “heat shock proteins”.
A precise, stable and, in particular, homogeneous heating of cell incubation vessels (especially when using multi-well specimen) is thus a major factor for a successful cell observation on the microscope. This is one of the main targets in the development of our products.
Two different buzzwords for the two different approaches of maintaining the temperature of cell culture vessels on the microscope are found in the web: “Stage Top Incubator” for small incubation inserts that are just put into the stage opening and “Cage Incubator” for large chamber incubators, enclosing most parts of the microscope. We at PeCon do not use the term “Cage Incubator”, because in our opinion your cell and tissue cultures must not be kept in a “cage” to prevent them from escaping.
PeCon offers a large selection of stage-top incubation products and accessories to enhance system’s performance for a precise, stable and uniform heating of specimen.
The “gold standard” for live cell imaging on the microscope is a large chamber incubator, that heats not only the specimen, but all microscope parts which are in contact with your sample. Additionally, mechanical drifts in the X, Y or Z-Axis are minimized, because the origin of such a material expansion of stages etc. by changing temperatures is reduced.
A large chamber incubator is also the basis of a very versatile and upgradeable incubation system. Transparent, light tight and laser-safe incubators are available.
pH value (CO2)
The optimal pH value for most cell lines is 7.4, but can be slightly different from cell type to cell type. Cell cultivation media comprise glucose, salts, amino acids, vitamins, other substances and a buffer substance to balance the pH value.
HEPES is a so-called “stable buffer”, it also maintains the pH value at ambient air conditions, however, this substance influences the cell behavior negatively. Even phototoxic effects are reported, which exclude its usage for microscopic cell observation.
The physiological buffer is NaHCO3, because it is the substance in most organisms to balance the pH value in blood and tissues. For this reason, NaHCO3 is used to buffer the pH of common cell culture media (like MEM, DMEM, RPMI 1640). Since this substance can dissociate into H2O and CO2, and the latter can just escape into the ambient air with its low CO2 concentration, the buffer is considered as “not stable”.
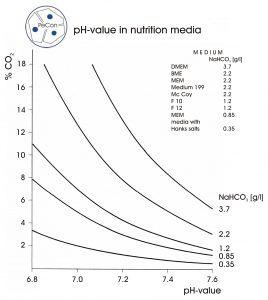
Therefore, the CO2 concentration in the atmosphere around the culture media has to be increased, until a steady state condition is achieved, where the same amount of CO2 escapes from and dissolves into the media. Depending on the NaHCO3 concentration used in media formulations, a CO2 concentration of 5.0% (2.2 g/l) or 7.5% (3.7 g/l) is needed.
A stable CO2 concentration ensures a stable pH in the nutrition media. Again, a homogeneous distribution in the incubation chamber is vital for mult well specimens. Our gas controllers, together with our small incubators or local CO2-Covers, generate this stable and uniform CO2 concentration. All controllers work with standard low-priced technical CO2 and do not need an additional compressed air supply. An in-house developed CO2 sensor ensures a fast and reliable measurement with a large detection range of 0-20% and a high resolution of 0.01%. It is specifically designed for cell cultivation needs, and is the basis for a quick and stable CO2 control with our controllers.
Oxygen (o2)
Cells under in vivo conditions are normally not exposed to the high oxygen level found in our atmosphere (20.8%), but are usually at an oxygen concentration of 10-13% (human blood) or lower. It is also known, that some differentiation processes like red blood cells from stem cells (erythropoiesis) only occur under lower oxygen concentrations. Higher oxygen concentrations are the trigger for the migration of special cells for performing wound healing.
Besides that, oxygen is a bi-radical, reacting very easily with other molecules and, thus, modifies them. Oxidative stress can lead to neurodegenerative diseases like Parkinson’s disease or Alzheimer’s disease. Oxygen also damages the DNA and cells have to perform elaborate DNA repair mechanisms to maintain their genome stability.
In order to offer cells the best possible conditions similar to in vivo, not only in the laboratory incubator, but also on the microscope, a reduction of the oxygen concentration should be considered. Our gas controllers for oxygen use an oxygen sensor with a very long lifetime and can reduce the oxygen concentration from ambient 20.8% down to 0.1% by a displacement with nitrogen. Due to their fast response times on setpoint changes, they can also be used for dynamic experiments.
Humidity
Evaporation of water out of the cell culture media happens every time when the relative humidity (rH) in the atmosphere around the cell culture vessel is <100%. However, most vessels have lids with only small gaps, so the drying effect is reduced, thus not eliminated. But cultivation times of several hours without a humidification of the atmosphere is possible without any negative effects on cell behavior or vitality.
The relative humidity is referenced to the amount of water that the air can hold at a given temperature. The higher the temperature, the more water can be absorbed. Therefore, when heating up the ambient air at 22°C with a relative humidity of 60% to 37°C, the relative humidity drops down to ~25%. While at 22°C only 19.4 g/m³ water is needed to have 100% rH, at 37°C already 43.8 g/m³ of water (more than twice the amount) is necessary.
Inside a large incubator, a humidification is easy. Just place the humidification bottle that is supplied with every PeCon gas controller and the supply tubing to the CO2-Cover inside the heated incubator. Physics will do its job and you will obtain a very effective humidification of the atmosphere around your specimen. For stage top systems, we have an external heater for the humidification flask. Combined with an isolated, heated tube, this is also very effective against the evaporation.
Since most cell culture media have to be changed after 2-3 days because they have been used up by the cells, this necessary media change resets the negative effects of evaporation. If, within this period of time, no harmful evaporation takes place, there is no need for extensive measures against the evaporation.
PeCon offers two different approaches to reduce the evaporation which can also be combined:
- Sealing the cultivation vessel with watertight but gas-permeable foils (FoilCovers)
- Increasing the humidity around the specimen (local humidification)


